|
Einstein and
Quantum Mechanics - Part 1
a
World
Without Einstein
Series
Welcome to the bizarre
micro-world of quantum mechanics. We explore Einstein's contributions
to and rejection of this pillar of 20th century physics.
Click here for Broadcast.
After
you have enjoyed the broadcast, get the book! All of the information
presented here, and more, can be found in World
without Einstein
|  |
|
|
Image
#1
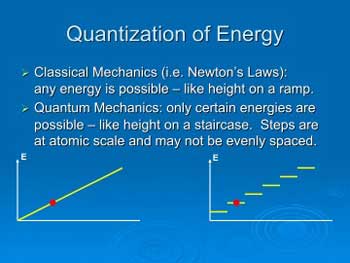
In the micro-world, nature is often “quantized”
like steps on a staircase rather than the ramp-like behavior of our
macro-world.
|
|
|

|
Image
#2
Thermodynamics
said hot bodies emit the same energy at all frequencies, but that made
the total energy infinite. Planck suggested a trick to prevent the
“Ultraviolet Catastrophe”: radiation must be quantized.
|
|
Image
#3
Before
QM, physics couldn’t explain why atoms didn’t collapse
|

|
|
|
Image
#4
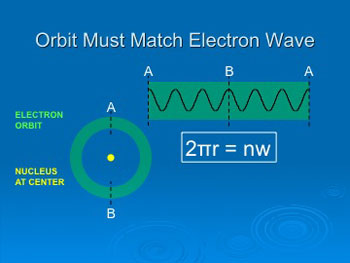
Particle-wave duality explains why atoms are
stable: electron orbits must contain integral numbers of wavelengths,
so they can’t spiral in closer than orbit #1.
|
|
Image
#5
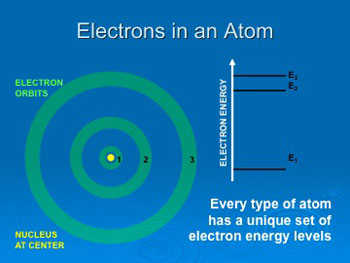
Electrons
can only occupy specific orbits with specific energies that are
different for each type of atom.
|
|
|
Image
#6
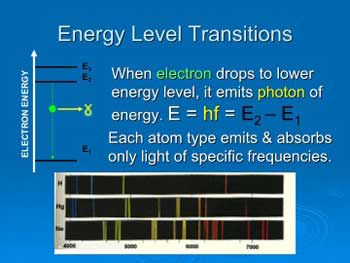
Atoms can emit or absorb only specific
energies equal to the difference in electron energies. These sets of
energies form a unique “spectrum” for each type of atom—a unique
fingerprint allowing us to measure the composition of stars.
|
|
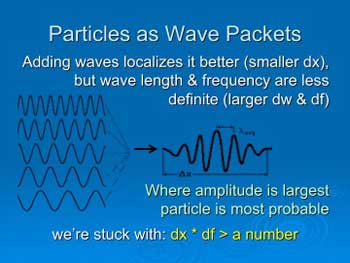
Image
#7
The waves on the left each have a definite wave
length and frequency, but indefinite locations—they are spread
everywhere. Adding waves of different frequencies forms a wave packet
with a location uncertainty dx and a frequency uncertainty df. Making
dx smaller makes df be larger, and vice versa—it’s a tradeoff.
|
|
|